The Role of Signaling Networks in Cell Migration
ABSTRACT
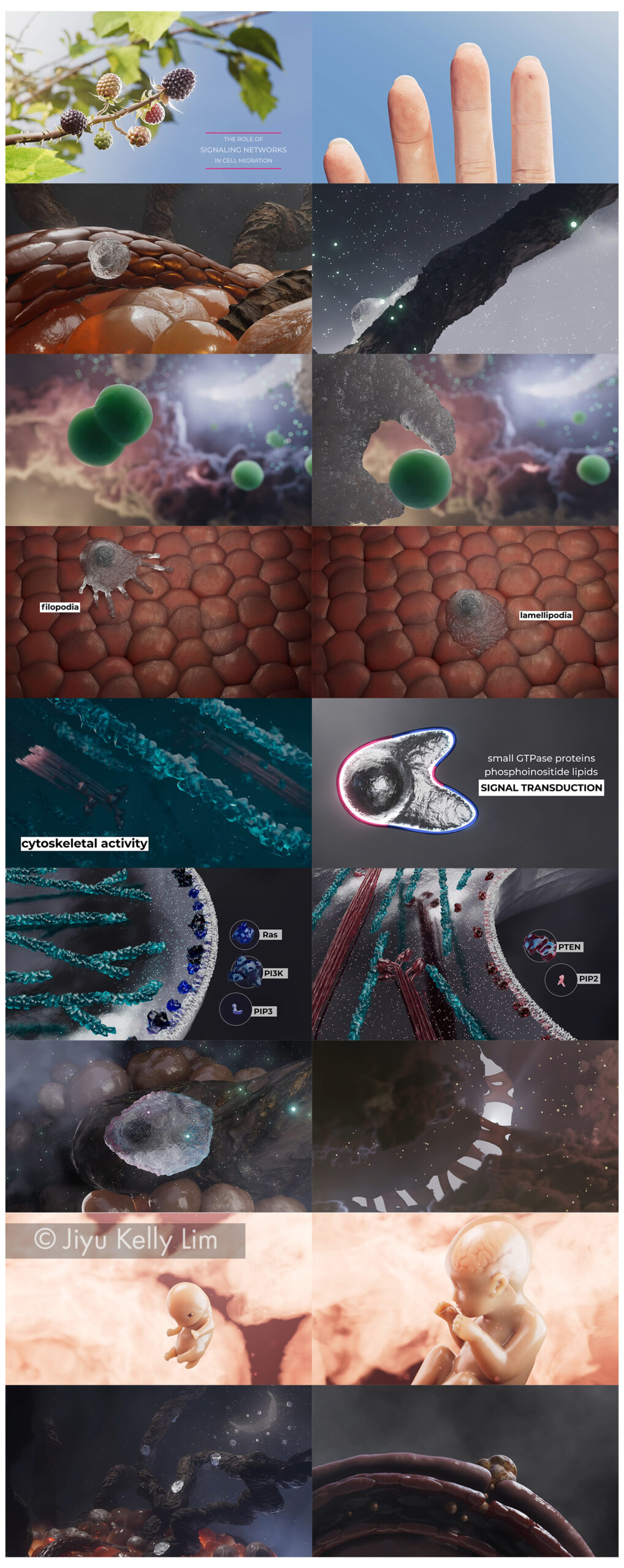
Life is dynamic. Cells are constantly changing shape. Many do so by displaying a variety of protrusions that not only vary their appearance but also play a key role in important cellular activities such as cell migration, division, and phagocytosis. These protrusions manifest in unique shapes and sizes, ranging from finger-like filopodia to sheet-like lamellipodia.
It is well known that these protrusions drive outward from the cell body by a combination of actin polymerization and actomyosin-based contractions, referred to as “cytoskeletal activity”. However, what determines the shape, and hence the identity of the protrusions, has remained a mystery until recently.
In recent years, a research team in the Johns Hopkins University Department of Cell Biology has making discoveries in a cell motility mechanism called Signal Transduction Excitable Network (STEN). STEN, a signaling network consisting of receptors, small GTPase proteins, and phosphoinositide lipids, determines the locations and lateral dimensions of cellular protrusions. Without STEN, cytoskeletal activity only produces transient, small extensions, or “puncta” which are ineffective in moving or reshaping cells. Manipulating the signal network can lead to alterations of the cytoskeletal system and morphing of the shape of the cell. Increasing or decreasing signal transduction activity can elevate or decrease the speed and range of wave propagation respectively, converting pseudopodia into wider lamellipodia, or narrower filopodia.
This novel finding provides a direction for future biomedical research as it shows STEN plays a critical role in cell migration and morphology, and dysregulation of this system can lead to the development of a variety of diseases including cancer, and developmental and metabolic abnormalities.
However, the mechanism of STEN is difficult to succinctly explain due to its three-dimensional, dynamic nature. Current teaching materials are limited to simple line diagrams and crude confocal microscopy videos and photographs, none of which are adequate to allow for in-depth understanding of this intricate process.
To solve this challenge, I propose a narrative 3D animation that can help learners visualize and comprehend this complex mechanism.
STORYBOARD

MATERIALS & METHODS

RESULTS